Technologies
Vascular Access Graft
InnAVasc’s novel graft modification technology is designed to create one contiguous flow lumen without transition points. The key innovative features of this technology include:
- Self-sealing needle stick zone which allows for durable, injury-proof cannulation and immediate use for hemodialysis post implantation
- Dialysis catheter reduction and elimination
- Error proof cannulation which allows for predictable, reliable access every time
- Ease of use regardless of cannulation experience, including patients, family members, and new dialysis technicians
The InnAVasc AV Graft is for investigational use only. The device has not been cleared to market by the U.S. FDA.
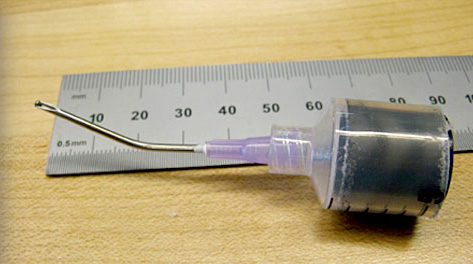
Bent needle after cannulation testing.
Chamber back plate can withstand extreme force from sharp needles without penetration.
Angiogram immediately post graft cannulation.
No extravasation (leakage of blood) from Bullet Proof graft (left), severe extravasation from standard graft (right).
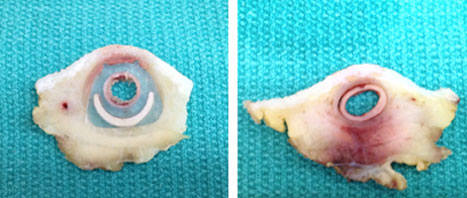
Cross section of Bullet Proof graft.
Cross section of Bullet Proof graft (left) and standard graft (right). Note severe inflammation and hematoma below posterior wall of standard graft and none below Bullet Proof.